|
| "Enjoya" pepper; marketing photo from the TwitterVerse. |
There was no information available about the genetics of the trait, as there had been no academic literature published on the new variety. Gardeners with the habit of growing their own plants from seed took this as a challenge. People around the globe independently said, "Can I can grow seeds from that pepper and get striped fruit in my garden?" Seeds were collected by those who found the peppers in their grocers and then shared via online forums to those who had not yet found them. Soon after, there were many little green seedlings being tended to around the world.
 |
| Typical flowers and fruit. |
Months later, the first reports on the plants started coming in. The plants were producing large bell peppers, but they were all ripening yellow. (I have reports of 11 plants maturing to produce yellow fruit.) As these reports were posted to the forums, interest in the plants waned. (Dreams of crossing the trait into jalapenos and other hot peppers quietly died.) If the amazing red flames weren't going to reappear, then why would anyone want to be growing these plants?
Where did these peppers come from?
The marketing site for the pepper says:
Now, 30 years later, nature has once again surprised us with a natural variation: the red/yellow striped pepper. In 2013, Wilfred van den Berg found this beautiful variety in his greenhouse in Est.But the US patent applied for the pepper says:
[0011] `E20B3751` was discovered in a screening trial of mutants of pepper variety `Maduro` conducted at Est, Netherlands. The mutant `E20B3751` was selected based on its vertical red and yellow stripes color and propagated vegetatively (i.e., asexually).I strongly suspect those responsible for writing the marketing site didn't want to say the variety was the result of a mutation breeding project in a high-tech lab, as such things tend to get a lot of people suspicious about their foods. This is only a slight fib, since the mutated variety is a variation of the natural pepper.
What draws my attention more is that the patent doesn't say anything at all about how the pepper plant was produced (aside from the general concept of a mutagenesis screen). The entirety of the patent starting on line [0046] is simply a rehashing of general plant biology and breeding. None of that tells us anything at all about the origin of the striped peppers. This is strongly counter to the basic idea of what patents are supposed to be. The earlier paragraphs of the patent do give a concise description of what the pepper is, as well as a listing of specific traits associated with it, so it isn't entirely a useless document.
Since there isn't any academic research published on the pepper and neither the patent or marketing information provide any biological details, we're going to have to see what we can figure out from basic principles.
Mutations in genes typically produce traits which are either dominant or recessive. (There are a few other scenarios, but we're not going to worry about them for now.) If the striped trait is recessive, then essentially all of the next generation would also have the trait.
If the striped trait was dominant, then [with perfect selfing] the next generation might all have the trait, but there are other scenarios. If the Enjoya pepper plant (remember, from the patent they are propagated assexually and so are all from the same genetic plant) was heterozygous for the dominant trait, then half of the next generation would remain heterozygous and have the trait. Another quarter would be homozygous for the no-stripes trait and the remaining plants would be homozygous for the striped trait. Dominant traits can sometimes also have recessive lethal characteristic, though it is rare. All together, at the very least 66.6% of the next generation should have stripes if the trait was due to a dominant nuclear mutation.
In either scenario, we should have the majority of the next generation with stripes. What do we see? Between my plants and those reported by other growers, we have 16 plants that have ripened fruit. All of which matured to yellow with no red stripes. This would be a very unexpected result for either model discussed above.
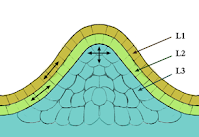 |
| Meristem figure from Wikipedia. |
 |
| Photo cropped from one at link. |
It looks like some of the red core cells are able to migrate to the surface of the fruit during early development. This results in the red stripes as the fruit then expands in size.
Since the red color is carried in tissue which isn't made into eggs or seeds, it appears unlikely that the seed-grown progeny of an Enjoya pepper would produce red or striped fruit.
Sorry folks, I think the game is up. We probably won't be able to breed flame-colored jalapenos. At least we've learned something about the biology of these peppers.
That the striped trait can't be passed down through seeds tells us something about the experiments which led to the Enjoya pepper. The patent indicates it came from a mutagenesis experiment, but gives no details. One of the easiest ways to do it would have been to soak a large batch of seeds in a chemical mutagen (like EMS) and then grow them out after treatment. EMS is relatively easy to work with and it would produce point mutations all over the nuclear and cytoplasmic genomes. I bet when that first plant matured its first fruit, there were amazed expressions all around.
The classical story of pepper color genetics (described at the-biologist-is-in.blogspot.ca/2015/11/the-color-of-peppers-2.html) suggests it would take two separate mutations to produce the rich yellow color seen in the Enjoya pepper. However, there are a lot of mutations which impact pepper color that don't really seem to fit the classical story. I strongly suspect the visible difference between the red and yellow fruit tissues is down to one mutation.
However, EMS is not something that would be used to make a single point mutation. It would instead create hundreds or thousands of point mutations per seed in this sort of mutagenesis experiment. Selection of the resulting progeny, as well as backcrossing to the parent type, would normally be used to clean up any unwanted deletarious mutations... but the striped trait would not have survived this process.
This means that the genome of the Enjoya pepper is probably chock-full of other potentially interesting mutations. Many of those mutations will be recessive and so only become visible in the second generation after treatment. The plants we've been growing from saved seed represent that second generation (referred to in shorthand as M2).
 |
| Enjoya-M2 with a transient anthocyanin shoulder. |
 |
| Enjoya-M2 with color-marked flowers. Interesting stripes on the unripe fruit. |
Two of my seven plants produced distinctively different plants. This suggests there are indeed numerous hidden recessive mutations in the Enjoya pepper. The relatively large fruit I've been getting from these plants and the potential to find other novelty mutations means I'll probably be growing quite a few of these M2 plants in the coming years.
References:
- Marketspeak:
- www.enjoya.com/en/taste-of-nature
- www.hortidaily.com/article/17276/New-red-and-yellow-striped-pepper-is-named-Enjoya
- Patent: appft.uspto.gov/netacgi/nph-Parser?Sect1=PTO2&Sect2=HITOFF&p=1&u=%2Fnetahtml%2FPTO%2Fsearch-bool.html&r=1&f=G&l=50&co1=AND&d=PG01&s1=pepper.AB.&s2=striped.AB.&OS=ABST%2Fpepper%20AND%20ABST%2Fstriped&RS=ABST%2Fpepper%20AND%20ABST%2Fstriped
- Discussions:
- www.tomatoville.com/showthread.php?t=41113&page=10
- www.growingfoodsavingseeds.co.uk/forum/main-forum/sweet-peppers-and-chillies/6906-enjoya-peppers
- www.growingfoodsavingseeds.co.uk/forum/welcome/welcome/6307-hi-nicollas
- c2cpeppers.proboards.com/thread/3987/flame-enjoya-pepper
- en.wikipedia.org/wiki/Meristem
- en.wikipedia.org/wiki/Variegation
- en.wikipedia.org/wiki/Ethyl_methanesulfonate
- EMS mutagenesis in seeds:
- cals.arizona.edu/research/schumaker/pdfs/Kim%202005.pdf
- www.ncbi.nlm.nih.gov/pubmed/22485968
- file.scirp.org/pdf/AS_2014072414110807.pdf
- www.esalq.usp.br/tomato/Induced Mutagenesis and Natural Genetic Variation in Tomato 'Micro-Tom'.pdf
- www.imedpub.com/articles/gamma-rays-and-ems-induced-flower-color-and-seed-mutants-in-cowpeavigna-unguiculata-l-walp.pdf
- www.insa.nic.in/writereaddata/UpLoadedFiles/PINSA/Vol55B_1989_5and6_Art25.pdf
- the-biologist-is-in.blogspot.ca/2015/11/the-color-of-peppers-2.html